Silver Bullet August 5 - WHO's math error on aerosols, clinical trials hit the road, how to become a guinea pig
Silver Bullet Newsletter
A weekly update on all things Covid19. News, public health guidance, trends, breakthroughs, and thousands of scientific papers distilled down to what you need to know right now
This week, Phase 3 clinical trials are underway in earnest for several Covid-19 vaccine candidates. With tens of thousands of volunteers being enrolled at clinical sites around the world, many people--maybe you!--now have the opportunity to receive an experimental vaccine. Meanwhile, some wrinkles in Covid-19 science are being ironed out--such as a gigantic math error the CDC and WHO have apparently been making when it comes to aerosols. We also have some new modeling forecasts and updated information on the role children may play in spreading the virus.
Remdesivir continues its pattern of good results in so-so studies
Gilead Sciences reported an interim analysis of two ongoing clinical studies of remdesivir. In one study, patients received two doses of remdesivir. In a second study, researchers are monitoring outcomes in a group of patients who received standard of care treatment without remdesivir. The analysis found that odds for recovery were about 2-fold higher in the remdesivir group and mortality was lower. However, this was not a randomized, placebo-controlled study--the gold standard for drug studies.
The CDC, WHO are are wrong by the numbers on droplets and aerosols
Jose-Luis Jimenez, PhD, an aerosol scientist from the University of Colorado, Boulder, published a detailed criticism of a recent article in JAMA that argues aerosols are not an important transmission pathway for SARS-CoV2. Perhaps the most compelling point he makes is that the article’s authors make a fundamental math error regarding the size of droplets. The JAMA article states, “Droplets are classically described as larger entities (>5 um) that rapidly drop to the ground by force of gravity, typically within 3 to 6 feet of the source person.”
“However, the actual size of droplets that fall to the ground that quickly corresponds to sizes larger than 50 μm, so 10 times the size and 1000 (!) times the mass given in the article. This fundamental error has been repeated for decades in guidance from the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO) and in medical papers, despite the correct physics having been figured out by Wells in 1934 and the error having been pointed out many times by other scientists,” Jimenez writes. He debunks many common beliefs related to aerosols, for example, that long range transmission is absent in Covid-19, superspreading events are rare, or that fomites (surface transmission) could in any way explain high profile superspreading events like the Skagit Valley Chorale. Jimenez attempted to publish a “letter in reply” in JAMA, but was told that the publication would not accept any letters to the editor on its Covid-related content. His response is therefore published in Medscape.
Eli Lilly launches clinical trial road show
Eli Lilly is deploying a fleet of retrofitted RVs to test its antibody drug on-site at nursing homes. The goal is to test the drug in one of the highest risk groups--people living in long term care facilities. Clinical trials are typically run through major medical centers, but Eli Lilly will be setting up its RVs as mobile labs in order to reach its goal of enrolling 2400 residents and staff at skilled nursing facilities in hotspots around the country. A trailer truck will deliver clinical trial supplies, and additional staff will be provided to support study operations.
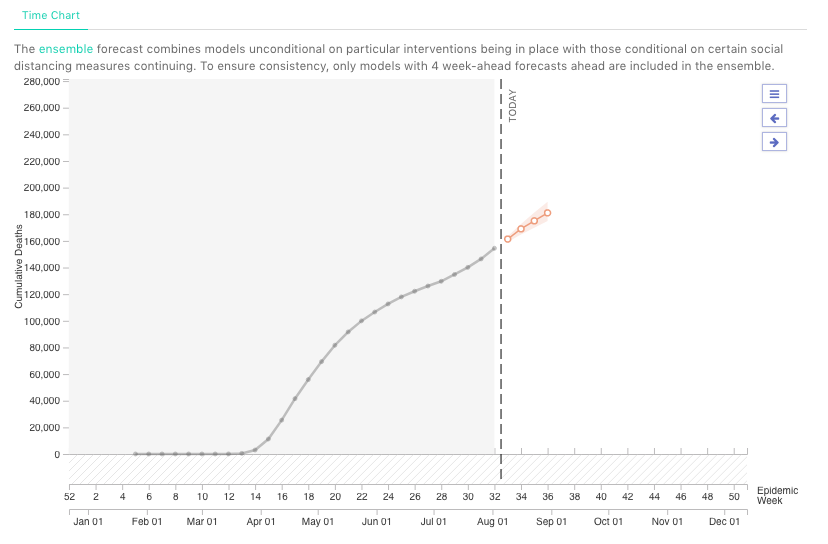
CDC predicts up 11,000 deaths a week by August 22
The CDC reports that an ensemble forecast from 32 modeling groups predicts weekly Covid-19 deaths will increase over the next month, rising to as many as 11,000 new deaths in the week ending Aug. 22, with as many as 182,000 cumulative US deaths. The forecast can be viewed by state or the US as a whole at the Covid-19 ForecastHub.
Modeling the effect of school opening on a pandemic ‘second wave’
British researchers modeled the outcome of two different school opening strategies under three different testing scenarios. Their results showed that an epidemic rebound could be prevented if at least 75% of people with symptoms are tested and 68% of contacts traced. If the numbers fell below that, reopening schools combined with relaxation of lockdown measures would set off a major second wave of infection more than twice the size of the original wave. The study is in The Lancet Child and Adolescent Health.
A reality check on vaccine development
A new analysis from the National Bureau of Economic Research highlights an inconvenient reality of vaccine development amid the race to develop a vaccine for Covid-19. The authors estimated the probability of success for clinical trials for vaccines infectious diseases. For industry sponsored programs, the overall odds of making it from Phase 1 clinical trials to approval successfully were 39.6%, and for non-industry sponsored programs, they were only 6.8%. (Some of this may be due to the fact that industry programs are chosen because they have a higher likelihood of success to begin with, whereas non-industry programs are often undertaken in response to an urgent public health need.) And in an op-ed for the New York Times, vaccine researcher Natalie Dean breaks down the hurdles that remain for all vaccine candidates in development. She points out that the currently available animal and early human data does not provide nearly enough evidence for safety and efficacy to approve the vaccine, or to begin distributing it before approval, especially in light of the fact that many vaccines with similarly promising early results have failed in large scale trials. “We must resist the desire to rush out a product. Creating vaccines is hard, and we should be prepared for the reality that some promising ones will not meet the FDA’s criteria. Researchers and the government should also commit to transparency so that people can see the results for themselves to understand the regulatory decisions,” writes Dean.
How to enroll in a clinical trial
One of the hurdles to developing a successful vaccine for Covid-19 is enrolling enough trial participants. Many of the companies developing vaccines are undertaking historically large trials in order to speed the process. Whereas a Phase 3 trial typically might enroll 1000 or 2000 subjects, some are enrolling as many as 30,000. Larger trials will theoretically produce faster results, but only if they get enough volunteers. If you want to help with the effort, you can search for a vaccine trial in your area at Clinicaltrials.gov. Entering “Covid-19” and “vaccine” in the search fields will get you a list of trials around the world. You can also narrow it down by region. If you are selected to participate, you will be given information about all of the risks of the trial so that you can make a decision about whether it’s right for you. You may be assigned to a placebo arm, so you can not assume you have any protection from the virus after receiving an injection. You can also search for clinical trials for drugs and other therapies for Covid-19 on the same site.
Pooled samples can reduce testing bottlenecks
By pooling samples into groups of 10, researchers were able to substantially reduce the number of tests needed to screen a group of 940 test samples that contained six random samples positive for SARS-CoV2. Instead of running 940 individual tests, they started with 94 pools of ten samples, and then ran a second round of 40 individual tests for the 4 pools of 10 that tested positive, for a total of 134 tests. In a cost analysis, the authors estimated that 1 million individuals can be tested for $9.1 million using pooled screening, compared to $58 million with standard screening. The study is published in The Journal of Molecular Diagnostics.
Transmission of Covid-19 in schools and day care ‘minimal’
An Australian study published in The Lancet carried out from January to April 2020 concludes that transmission of the coronavirus in educational settings was ‘minimal,’ and considerably less than that of other respiratory viruses like influenza, particularly between children and from children to adults.
Common cold immunity may offer some protection from new coronavirus
Memory helper T cells that recognize common cold coronaviruses also recognize some matching sites on the Covid-19 virus, SARS-CoV2. This may explain why some people have milder symptoms than others. That’s according to a study is in the Aug. 4 edition of Science.
Blood pressure drugs don’t increase risk
Hypertension is a major risk factor for severe illness in Covid-19. There has been concern that commonly used blood pressure drugs could lead to worse outcomes, since those drugs increase the number of ACE2 receptors that the coronavirus uses to enter cells. A new study in The Journal of Infectious Disease looked at more than 600 patients with hypertension and Covid-19, and concluded that patients taking ACE inhibitors and angiotensin II receptor blockers were much less likely to be admitted to the ICU than those who discontinued those medications.
In depth
How the pandemic defeated America by Ed Yong
A virus has brought the world’s most powerful country to its knees.
I’d need evidence before I’d get a Covid-19 vaccine. It doesn’t exist yet. by Natalie Dean
Currently available early stage data are not sufficient to prove a vaccine is safe and effective.
Scared that Covid-19 immunity won’t last? Don’t be by Akiko Iwasaki and Ruslan Medzhitov
Dropping antibody counts aren’t a sign that our immune system is failing against the coronavirus, nor an omen that we can’t develop a viable vaccine.
Be well!
--Catherine Shaffer

